Carbonic acid
| Carbonic acid | |
|---|---|
 |
|
 |
|
|
Other names
Carbon dioxide solution, dihydrogen carbonate (IUPAC), acid of air, aerial acid
|
|
| Identifiers | |
| CAS number | 463-79-6 |
| ChemSpider | 747 |
|
SMILES
O=C(O)O
|
|
|
InChI
InChI=1/CH2O3/c2-1(3)4/h(H2,2,3,4)
Key: BVKZGUZCCUSVTD-UHFFFAOYAU |
|
| Properties | |
| Molecular formula | H2CO3 |
| Molar mass | 62.03 g/mol |
| Density | 1.0 g/cm3 (dilute soln.) |
| Melting point |
n/a |
| Solubility in water | Exists only in solution |
| Acidity (pKa) | see acidity of carbonic acid |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) |
|
| Infobox references | |
- Not to be confused with carbolic acid, an antiquated name for phenol.
Carbonic acid is the organic compound with the formula H2CO3 (equivalently OC(OH)2). It is also a name sometimes given to solutions of carbon dioxide in water, which contain small amounts of H2CO3. The salts of carbonic acids are called bicarbonates (or hydrogen carbonates) and carbonates. It is a weak acid.
When dissolved in water, carbon dioxide exists in equilibrium with carbonic acid:
- CO2 + H2O
 H2CO3
H2CO3
The hydration equilibrium constant at 25 °C is Kh = [H2CO3]/[CO2] = 1.70×10−3: hence, the majority of the carbon dioxide is not converted into carbonic acid, remaining as CO2 molecules. In the absence of a catalyst, the equilibrium is reached quite slowly. The rate constants are 0.039 s−1 for the forward reaction (CO2 + H2O → H2CO3) and 23 s−1 for the reverse reaction (H2CO3 → CO2 + H2O). Carbonic acid is used in the making of soft drinks, inexpensive and artificially carbonated sparkling wines, and other bubbly drinks. The addition of two equivalents of water to CO2 would give orthocarbonic acid, C(OH)4, which is unimportant in aqueous solution.
Contents |
Role of carbonic acid in blood
Carbonic acid is an intermediate step in the transport of CO2 out of the body via respiratory gas exchange. The hydration reaction of CO2 is generally very slow in the absence of a catalyst, but red blood cells contain carbonic anhydrase, which both increases the reaction rate and dissociates a hydrogen ion (H+) from the resulting carbonic acid, leaving bicarbonate (HCO3-) dissolved in the blood plasma. This catalysed reaction is reversed in the lungs, where it converts the bicarbonate back into CO2 and allows it to be expelled. This equilibration plays an important role as a buffer in mammalian blood.
Role of carbonic acid in ocean chemistry
The oceans of the world have absorbed almost half of the CO2 emitted by humans from the burning of fossil fuels.[1] The extra dissolved carbon dioxide has caused the ocean's average surface pH to shift by about 0.1 unit from pre-industrial levels.[2] This process is known as ocean acidification.
Acidity of carbonic acid
Carbonic acid is diprotic: it has two protons, which may dissociate from the parent molecule. Thus there are two dissociation constants, the first one for the dissociation into the bicarbonate (also called hydrogen carbonate) ion HCO3−:
- H2CO3
 HCO3− + H+
HCO3− + H+ - Ka1 = 2.5×10−4 ; pKa1 = 3.60 at 25 °C.
This indicates that H2CO3 is a stronger acid than acetic acid and formic acid. Its high acidity reflects the influence of the electronegative oxygen substituent.
The second for the dissociation of the bicarbonate ion into the carbonate ion CO32−:
- HCO3−
 CO32− + H+
CO32− + H+ - Ka2 = 5.61×10−11 ; pKa2 = 10.33 at 25 °C and Ionic Strength = 0.0.
Care must be taken when quoting and using the first dissociation constant of carbonic acid. In aqueous solution carbonic acid only exists in equilibrium with carbon dioxide, and the concentration of H2CO3 is much lower than the dissolved CO2 concentration. Since it is not possible to distinguish between H2CO3 and dissolved CO2 (referred to as CO2(aq)) by conventional methods, H2CO3* is used to represent the two species when writing the aqueous chemical equilibrium equation. The equation may be rewritten as follows (c.f. sulfurous acid):
- H2CO3*
 HCO3− + H+
HCO3− + H+ - Ka = 4.6×10−7(General Chemistry: An Integrated Approach Third Edition); pKa = 6.352 at 25 °C and Ionic Strength = 0.0.(NIST CRITICAL Database)
Whereas this pKa is quoted as the dissociation constant of carbonic acid, it is ambiguous: it might better be referred to as the acidity constant of dissolved carbon dioxide, as it is particularly useful for calculating the pH of CO2-containing solutions.
pH and composition of a carbonic acid solutions
At a given temperature, the composition of a pure carbonic acid solution (or of a pure CO2 solution) is completely determined by the partial pressure 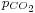 of carbon dioxide above the solution. To calculate this composition, account must be taken of the above equilibria between the three different carbonate forms (H2CO3, HCO3− and CO32−) as well as of the hydration equilibrium between dissolved CO2 and H2CO3 with constant
of carbon dioxide above the solution. To calculate this composition, account must be taken of the above equilibria between the three different carbonate forms (H2CO3, HCO3− and CO32−) as well as of the hydration equilibrium between dissolved CO2 and H2CO3 with constant ![\scriptstyle K_h=\frac{[H_2CO_3]}{[CO_2]}](/2010-wikipedia_en_wp1-0.8_orig_2010-12/I/548164e9dafca2231c39541c7e511fa1.png) (see above) and of the following equilibrium between the dissolved CO2 and the gaseous CO2 above the solution:
(see above) and of the following equilibrium between the dissolved CO2 and the gaseous CO2 above the solution:
- CO2(gas)
 CO2(dissolved) with
CO2(dissolved) with ![\scriptstyle \frac{[CO_2]}{p_{CO_2}}=\frac{1}{k_\mathrm{H}}](/2010-wikipedia_en_wp1-0.8_orig_2010-12/I/f39b32d825fae72e1bdf768f57e48327.png) where kH=29.76 atm/(mol/L) at 25°C (Henry constant)
where kH=29.76 atm/(mol/L) at 25°C (Henry constant)
The corresponding equilibrium equations together with the ![\scriptstyle[H^+][OH^-]=10^{-14}](/2010-wikipedia_en_wp1-0.8_orig_2010-12/I/820d8be050a0a727f6597d89d086a9e9.png) relation and the charge neutrality condition
relation and the charge neutrality condition ![\scriptstyle[H^+]=[OH^-]+[HCO_3^-]+2[CO_3^{2-}]](/2010-wikipedia_en_wp1-0.8_orig_2010-12/I/8799cc0efd127b08f6494b6015e9bc97.png) result in six equations for the six unknowns [CO2], [H2CO3], [H+], [OH−], [HCO3−] and [CO32−], showing that the composition of the solution is fully determined by
result in six equations for the six unknowns [CO2], [H2CO3], [H+], [OH−], [HCO3−] and [CO32−], showing that the composition of the solution is fully determined by 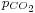 . The equation obtained for [H+] is a cubic whose numerical solution yields the following values for the pH and the different species concentrations:
. The equation obtained for [H+] is a cubic whose numerical solution yields the following values for the pH and the different species concentrations:
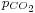 (atm) |
pH | [CO2] (mol/L) |
[H2CO3] (mol/L) |
[HCO3−] (mol/L) |
[CO32−] (mol/L) |
|---|---|---|---|---|---|
| 10−8 | 7.00 | 3.36 × 10−10 | 5.71 × 10−13 | 1.42 × 10−9 | 7.90 × 10−13 |
| 10−7 | 6.94 | 3.36 × 10−9 | 5.71 × 10−12 | 5.90 × 10−9 | 1.90 × 10−12 |
| 10−6 | 6.81 | 3.36 × 10−8 | 5.71 × 10−11 | 9.16 × 10−8 | 3.30 × 10−11 |
| 10−5 | 6.42 | 3.36 × 10−7 | 5.71 × 10−9 | 3.78 × 10−7 | 4.53 × 10−11 |
| 10−4 | 5.92 | 3.36 × 10−6 | 5.71 × 10−9 | 1.19 × 10−6 | 5.57 × 10−11 |
| 3.5 × 10−4 | 5.65 | 1.18 × 10−5 | 2.00 × 10−8 | 2.23 × 10−6 | 5.60 × 10−11 |
| 10−3 | 5.42 | 3.36 × 10−5 | 5.71 × 10−8 | 3.78 × 10−6 | 5.61 × 10−11 |
| 10−2 | 4.92 | 3.36 × 10−4 | 5.71 × 10−7 | 1.19 × 10−5 | 5.61 × 10−11 |
| 10−1 | 4.42 | 3.36 × 10−3 | 5.71 × 10−6 | 3.78 × 10−5 | 5.61 × 10−11 |
| 100 | 3.92 | 3.36 × 10−2 | 5.71 × 10−5 | 1.20 × 10−4 | 5.61 × 10−11 |
| 2.5 × 100 | 3.72 | 8.40 × 10−2 | 1.43 × 10−4 | 1.89 × 10−4 | 5.61 × 10−11 |
| 101 | 3.42 | 3.36 × 10−1 | 5.71 × 10−4 | 3.78 × 10−4 | 5.61 × 10−11 |
- We see that in the total range of pressure, the pH is always largely lower than pKa2 so that the CO32− concentration is always negligible with respect to HCO3− concentration. In fact CO32− plays no quantitative role in the present calculation (see remark below).
- For vanishing
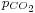 , the pH is close to the one of pure water (pH = 7) and the dissolved carbon is essentially in the HCO3− form.
, the pH is close to the one of pure water (pH = 7) and the dissolved carbon is essentially in the HCO3− form. - For normal atmospheric conditions (
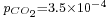 atm), we get a slightly acid solution (pH = 5.7) and the dissolved carbon is now essentially in the CO2 form. From this pressure on, [OH−] becomes also negligible so that the ionized part of the solution is now an equimolar mixture of H+ and HCO3−.
atm), we get a slightly acid solution (pH = 5.7) and the dissolved carbon is now essentially in the CO2 form. From this pressure on, [OH−] becomes also negligible so that the ionized part of the solution is now an equimolar mixture of H+ and HCO3−. - For a CO2 pressure typical of the one in soda drink bottles (
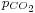 ~ 2.5 atm), we get a relatively acid medium (pH = 3.7) with a high concentration of dissolved CO2. These features contribute to the sour and sparkling taste of these drinks.
~ 2.5 atm), we get a relatively acid medium (pH = 3.7) with a high concentration of dissolved CO2. These features contribute to the sour and sparkling taste of these drinks. - Between 2.5 and 10 atm, the pH crosses the pKa1 value (3.60) giving a dominant H2CO3 concentration (with respect to HCO3−) at high pressures.
Remark
- As noted above, [CO32−] may be neglected for this specific problem, resulting in the following very precise analytical expression for [H+]:
![\scriptstyle[H^+] \simeq \left( 10^{-14}+\frac {K_hK_{a1}}{k_\mathrm{H}} p_{CO_2}\right)^{1/2}](/2010-wikipedia_en_wp1-0.8_orig_2010-12/I/831ffd4f77d667186b1d24cf7131235e.png)
Spectroscopic studies of carbonic acid
Theoretical calculations show that the presence of even a single molecule of water causes carbonic acid to revert to carbon dioxide and water. In the absence of water, gaseous carbonic acid is predicted to be stable with a half-life of 180,000 years.[3]
It has long been recognized that it is impossible to obtain pure carbonic acid at room temperatures (about 20 °C or about 70 °F). It can be generated by exposing a frozen mixture of water and carbon dioxide to high-energy radiation, and then warming to remove the excess water. The carbonic acid that remained was characterized by infrared spectroscopy. The fact that the carbonic acid was prepared by irradiating a solid H2O + CO2 mixture has given rise to suggestions that H2CO3 might be found in outer space, where frozen ices of H2O and CO2 are common, as are cosmic rays and ultraviolet light, to help them react.[3] The same carbonic acid polymorph (denoted beta-carbonic acid) was prepared by heating alternating layers of glassy aqueous solutions of bicarbonate and acid in vacuo, which causes protonation of bicarbonate, followed by removal of the solvent. Alpha-carbonic acid was prepared by the same technique using methanol rather than water as a solvent.
See also
References
- Welch, M. J.; Lipton, J. F.; Seck, J. A. (1969). "Tracer studies with radioactive oxygen-15. Exchange between carbon dioxide and water". J. Phys. Chem. 73 (335): 3351. doi:10.1021/j100844a033.
- Jolly, W. L. (1991). Modern Inorganic Chemistry (2nd Edn.). New York: McGraw-Hill. ISBN 0-07-112651-1.
- Moore, M. H.; Khanna, R. (1991). "Infrared and Mass Spectral Studies of Proton Irradiated H2O+Co2 Ice: Evidence for Carbonic Acid Ice: Evidence for Carbonic Acid". Spectrochimica Acta 47A: 255–262. doi:10.1016/0584-8539(91)80097-3.
- W. Hage, K. R. Liedl; Mayer, E.; Hallbrucker, A; Mayer, E (1998). "Carbonic Acid in the Gas Phase and Its Astrophysical Relevance". Science 279 (5355): 1332–1335. doi:10.1126/science.279.5355.1332. PMID 9478889.
- Hage, W.; Hallbrucker, A.; Mayer, E. (1993). "Carbonic Acid: Synthesis by Protonation of Bicarbonate and Ftir Spectroscopic Characterization Via a New Cryogenic Technique". J. Am. Chem. Soc. 115: 8427–8431. doi:10.1021/ja00071a061.
- Hage, W.; Hallbrucker, A.; Mayer, E. (1995). "A Polymorph of Carbonic Acid and Its Possible Astrophysical Relevance". J. Chem. Soc. Farad. Trans. 91: 2823–2826. doi:10.1039/ft9959102823.
References
- ↑ Sabine, C.L.; et al. (2004). " "The Oceanic Sink for Anthropogenic CO2". Science 305 (5682): 367–371. doi:10.1126/science.1097403. PMID 15256665. http://www.sciencemag.org/cgi/content/short/305/5682/367".
- ↑ "Ocean Acidification Network". http://ioc3.unesco.org/oanet/FAQacidity.html.
- ↑ 3.0 3.1 Loerting, T.; Tautermann, C.; Kroemer, R.T.; Kohl, I.; Mayer, E.; Hallbrucker, A.; Liedl, K. R. (2001). "On the Surprising Kinetic Stability of Carbonic Acid". Angew. Chem. Int. Ed. 39: 891–895. doi:10.1002/(SICI)1521-3773(20000303)39:5<891::AID-ANIE891>3.0.CO;2-E.
External links
- Ask a Scientist: Carbonic Acid Decomposition
- Why was the existence of carbonic acid unfairly doubted for so long?
- Carbonic acid/bicarbonate/carbonate equilibrium in water: pH of solutions, buffer capacity, titration and species distribution vs. pH computed with a free spreadsheet
- How to calculate concentration of Carbonic Acid in Water
|
|||||||||||